Exosome-Depleted UltraGRO™-PURE GI Research grade
Product Type:Exosome-Depleted, Gamma irradiated FDhPL
Attribute:Xeno-free
Grade:Research grade
Product Name:Exosome-Depleted UltraGRO™-PURE GI
Catalog Number:HPCHEFRLI50, HPCHEFRLI10, HPCHEFRLI05
-
Description:
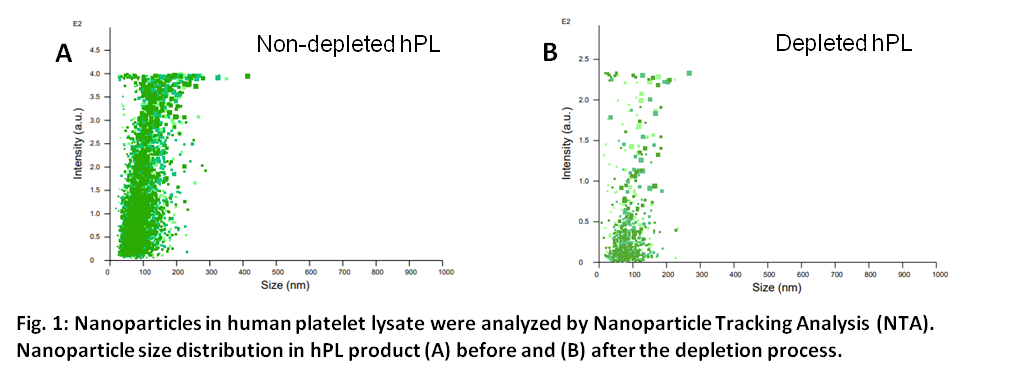
Exosome-Depleted UltraGRO™-PURE GI cell culture supplement is a gamma irradiated viral inactivated, exosome-depleted, xeno-free to support cell culture, especially for exosome secretion and isolation assay from research and clinical trials to commercial applications. More than 95% of nanoparticles have been removed from the original human platelet lysate, allowing target cells to produce functional exosomes and other cell-derived extracellular vesicles for further purification, analysis, and therapeutic purposes.
-
Quality Control Test:
1. Appearance: Slight yellow
2. Endotoxin: ≤ 10 EU/mL
3. Sterility: No Growth
4. pH: 6.5 - 8.5
5. Particle depleted rate: ≧ 95%
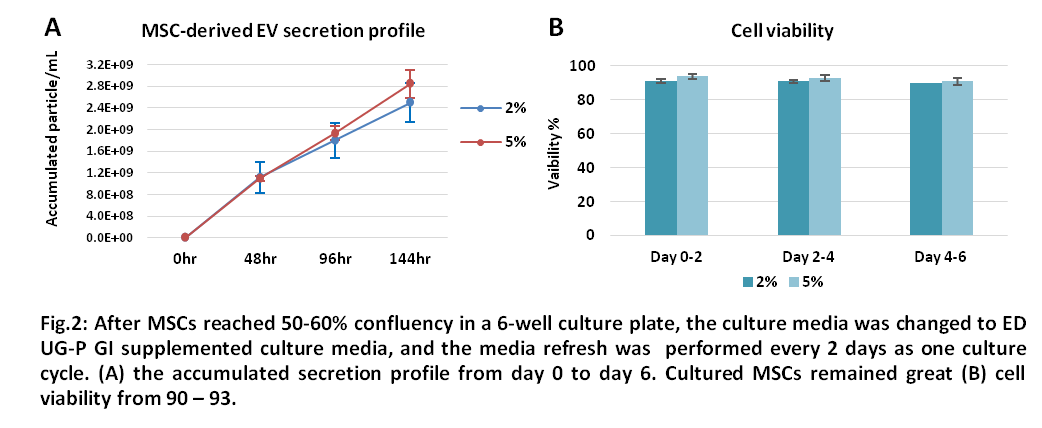
6. Cell assay (Viability): MSC viability ≧ 80%
7. Gamma Irradiation Dose: 25~40 kGy
-
Storage and Shipping Information:
Exosome-Depleted UltraGRO™-PURE GI is most stable when stored frozen until needed. The recommended storage temperature is -20°C. Thaw frozen Exosome-Depleted UltraGRO™-PURE GI product in 37°C water bath before use. Once Exosome-Depleted UltraGRO™-PURE GI is thawed, it is recommended to fully use for completed medium preparation (e.g., 5%) on the same day, or to divide it into single-use aliquots and store unused aliquots at -20°C. The product will be shipped with dry ice.
-
Packaging Information:
500mL, 100mL, 50mL plastic bottles
-
Media Form:
Frozen Liquid
-
Application:
For human cell culture in producing exosome and other cell-derived extracellular vesicles processing applications.
-
Instructions for Use:
1. Once the target cells reach desired cell confluency, Exosome-Depleted UltraGRO™-PURE GI supplemented culture media can be introduced to initiate cell-derived EV production.
2. We recommend applying with 2-5% (v/v) of supplemented media during the culture period. Perform conditioned media harvest and media refresh in every 2-3 days.
-
Cell Lines:
bone marrow mesenchymal stem cells, adipose tissue derived mesenchymal stem cells, umbilical cord derived mesenchymal stem cells, other mesenchymal stem cells
-
Important Information:
1. Insoluble particles may form in thawed Exosome-Depleted UltraGRO™- PURE GI. Published research has shown that particles will not alter the performance of the product. It is recommended to remove the precipitates by centrifuge at 3,400 xg for 3~5 minutes. Alternatively, applying 0.22 µm filtration to the prepared Exosome-Depleted UltraGRO™-PURE GI containing medium (e.g., 5%) will not affect the cell culture performance.
2. 0.22μm filtering is NOT recommended for the 100% Exosome-Depleted UltraGRO™-PURE GI.
3. Repeated freeze-thaw cycles should be avoided as they will cause an increase in insoluble precipitates.
-
Safety information:
1. Follow the handling instructions outlined in the Material Safety Date Sheets (MSDS). Wear appropriate protective eyewear, clothing, and gloves.
2. Exosome-Depleted UltraGRO™-PURE GI is a cell culture supplement collected from healthy donors at FDA licensed centers. Each donor has been tested using FDA licensed tests and found nonreactive for HBsAg, Hepatitis B core antibody (anti-HBc), HIV antibody (anti-HIV-1/2), Hepatitis C antibody (anti-HCV), HTLV-1/2 antibody (anti-HTLV-1/2), Trypanosoma cruzi antibody (anti-T.cruzi), HIV-1, HCV, HBV, WNV nucleic acid testing and Syphilis microhemagglutination test.
3. This product is for in vitro use only.
 Language
Language