UltraGRO™ GMP grade
Product Type:hPL
Attribute:Xeno-free
Grade:GMP grade
Product Name:UltraGRO™
Catalog Number:HPCPLCGL50, HPCPLCGL10, HPCPLCGL05
-
Description:
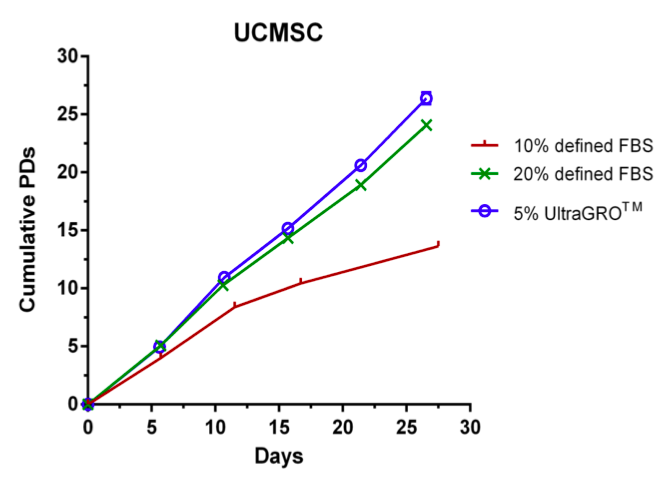
UltraGRO™ cell culture supplement is a xeno-free, animal serum-free, and heparin-requiring supplement for replacing FBS (fetal bovine serum) to support cell expansion from research and clinical trials to commercial application . UltraGRO™ contains abundant growth factors and cytokines necessary for research or industrial cell growth and proliferation of multiple cell types (e.g. MSCs) in research-scale or industrial-scale production.
-
Quality Control Test:
1. Appearance: Yellow to Buff liquid
2. Total Protein: 4.0 - 8.0 g/dL
3. pH: 6.5 - 8.5
4. Osmolality: 275 - 355 mOsm/Kg
5. Endotoxin: ≤ 10 EU/mL
6. Mycoplasma: Not Detected
7. Sterility: No Growth
8. Performance Test: Test and record
-
Storage and Shipping Information:
UltraGRO™ is most stable when stored frozen until needed. The recommended storage temperature is -20°C. Thaw frozen UltraGRO™ product in 37°C water bath before use. Once UltraGRO™ is thawed, it is recommended to fully use for completed medium preparation (e.g. 5%) the same day, or to divide it into single-use aliquots and store unused aliquots at -20°C.
-
Packaging Information:
500mL, 100mL, 50mL plastic bottles
-
Media Form:
Frozen Liquid
-
Application:
For human ex-vivo tissue and cell culture processing applications.
-
Instructions for Use:
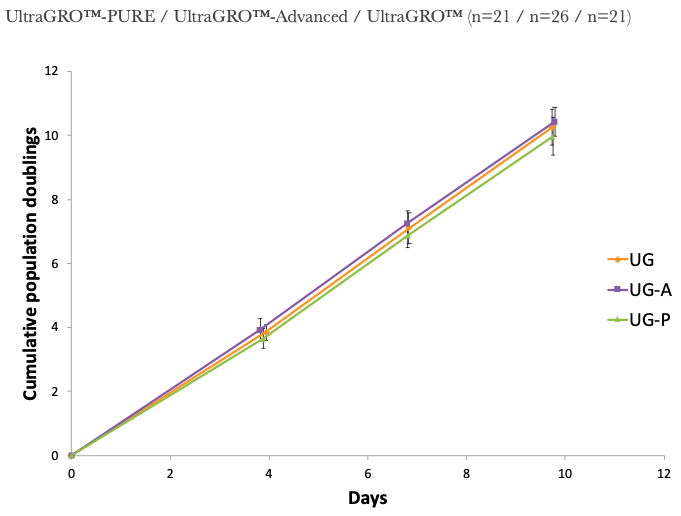
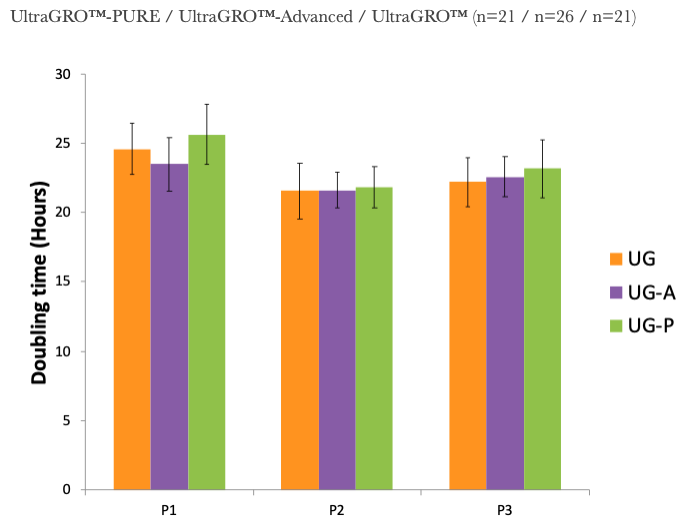
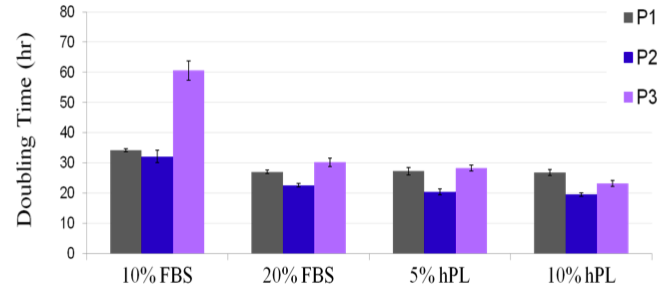
1. UltraGRO™ shows optimal growth of MSC at 5% (v/v) in typical cell culture media, i.e. α-MEM, which contains 2mM L-Glutamine as final concentrate.
2. We recommend seeding MSCs at approximately 3x103~6x103 per cm2.
3. UltraGRO™ requires heparin at a final concentration of 2 IU/mL to be added in the culture media when supplemented with 5% UltraGRO™. Failure to add heparin will result in coagulation during cell culture in typical media.
-
Cell Lines:
bone marrow mesenchymal stem cells, adipose tissue derived mesenchymal stem cells, umbilical cord derived mesenchymal stem cells, other mesenchymal stem cells
-
Important Information:
1. Clotting or insoluble particles may form in thawed UltraGRO™. Published research has shown that particles will not alter the performance of the product.
2. Clotting or insoluble particles may form in thawed UltraGRO™, it is recommended to centrifuge at 3,400 xg for 3~5 minutes or to filter the liquid concentrate with a sterile 40 μm Cell Strainer to remove insoluble particles.
3. Filtering the completed medium (e.g. 5%), after UltraGRO™ is diluted in the basal medium, will not affect UltraGRO™ supplemented cell culture performance. However, 0.22 μm filtering is NOT recommended for the 100% UltraGRO™ concentrate, as this may reduce 5% UltraGRO™ cell culture performance.
4. Repeated freeze-thaw cycles should be avoided as they will cause an increase in insoluble precipitates and resulting potential decrease in UltraGRO™ performance.
-
Safety information:
1. Follow the handling instructions outlined in the Material Safety Data Sheets (MSDSs). Wear appropriate protective eyewear, clothing, and gloves.
2. UltraGRO™ is a cell culture supplement derived from human platelets collected from healthy donors at FDA or HC licensed blood centers. Each donor has been tested using FDA/HC-licensed tests and found nonreactive for HBsAg, Hepatitis B core antibody (anti-HBc), HIV antibody (anti-HIV-1/2), Hepatitis C antibody (anti-HCV), HTLV-1/2 antibody (anti-HTLV-1/2), Trypanosoma cruzi antibody (anti-T. cruzi), HIV-1, HCV, HBV, WNV nucleic acid testing and Syphilis microhemagglutination test.
3. This product is manufactured, tested and released in compliance with the relevant GMP guidelines. This product is for in vitro use only.
 Language
Language